The post 2025 Autism Research Year in Review appeared first on Autism Science Foundation.
]]>2025 has been a monumentally challenging year for scientists in general and for autism researchers in particular, but despite drastic federal funding cuts, scientists continued to make progress toward understanding autism’s causes, improving diagnosis, and developing more effective treatments and supports for individuals with autism and their families. According to an Autism Science Foundation survey, about $80 million in autism funding was eliminated early this year as part of Department of Government Affairs (DOGE) cuts. This included approximately $80 million in autism research across the NSF, CDC, and NIH. In September, we received some encouraging news when the NIH announced $50 million in funding for 13 projects under the new Autism Data Science Initiative (ADSI), which will utilize U.S. datasets to investigate gene–environment interactions. These studies will examine a range of environmental factors, individually and in combination, and explore their roles in autism prevalence and causation.
Meanwhile, there was substantial progress toward understanding the biology of autism, including new insights into causes and effective supports, and important findings on biological subtyping.
Heterogeneity: Studies Confirm Autism Subtypes
One of the clearest scientific themes of 2025 was progress in understanding the differences of people with autism across the spectrum. Large-scale biological and behavioral analyses published this year identified reproducible subgroups within autism, including groups that differ in genetic backgrounds, biological features, changes in behavioral features over time, and co-occurring medical or behavioral profiles. These studies have the potential to shift the field away from treating autism as a single, uniform condition toward stratified, precision approaches and targeted supports.
Researchers used artificial intelligence paradigms to identify behavioral patterns and group individuals by shared features. They then compared biological characteristics across these groups to identify underlying mechanisms. This work began last year, and the findings reported in 2025 have been among the most impactful to date in defining autism subtypes. Using this approach, one study identified four subtypes, including one that closely aligns with profound autism (“broadly affected”), another characterized by higher rates of psychiatric conditions such as anxiety or ADHD (“moderately affected”), and two groups with milder challenges.1 Another study found that age at diagnosis—early childhood versus adolescence—was the strongest predictor of subtype. Children diagnosed earlier tended to have delays in language, motor skills, and overall development, while those diagnosed later were more likely to have OCD, ADHD, or anxiety.2 Those who are diagnosed later had co-morbid psychiatric issues that may have masked an autism diagnosis.
Genes are early determinants of biology and behavior, making it critical to understand how genetics contributes to these behavioral differences. These studies found that individuals with greater challenges tend to carry larger, rarer genetic variants, whereas those with milder or later-diagnosed autism show a higher burden of smaller, more common variants.1,2 Certain rare variants, known as de novo variants and not inherited from either parent, are associated with more severe autism features.3
Looking beyond genetics to brain structure, additional studies showed that the severity of autism traits measured by the ADOS, both in individuals with autism and in individuals with ADHD, was a primary driver of differences in brain structure, rather than diagnostic category alone.4 The overlap in behavioral features, brain structure, and genetics across autism and many psychiatric disorders highlights the transdiagnostic nature of many neurodevelopmental traits5,4 calling into question existing diagnostic boundaries. Many of the brain changes studied, including differences in cell density, were similar in individuals with ADHD and autism and more dependent on severity of symptoms.4,6 Other studies demonstrated that autism-related brain differences are also present in individuals without an autism diagnosis who nonetheless show elevated autism traits.7 Taken together, these studies confirm that autism is not a single condition. As research progresses, a key priority will be to better understand subgroups within the broader non-profound autism population so that supports and services can be more effectively tailored.
Genetics and Environmental Factors
Individual functioning is influenced not only by genetics but also by environmental factors unique to each individual or family. These include chemical, nutritional, and contextual influences that warrant further study.8 While some environmental factors, like prenatal birth, may affect the probability of an autism diagnosis independent of genetic risk9,10, they may also modify outcomes and capabilities in individuals who are already diagnosed.11,12 Scientists increasingly agree that a broad range of environmental exposures should be studied in relation to developmental outcomes across diagnostic boundaries and along a continuum, rather than focusing narrowly on autism alone.8,13
This year, studies also helped exonerate environmental factors that are not related to autism, including acetaminophen. In 2025, global health authorities and scientific reviews reaffirmed that there is no proven causal link between Tylenol (acetaminophen) use during pregnancy and autism. Organizations such as the World Health Organization and the American Academy of Pediatrics noted that existing studies show inconsistent associations and do not establish causation. These clarifications came amid political claims suggesting a connection, prompting experts and advocacy groups like ASF to emphasize that autism’s causes are complex and not attributable to anything parents did or did not do before, during, or after pregnancy.14
Sex Differences
Research in 2025 also advanced understanding of sex differences in autism, particularly the biological mechanisms that shape them. Large genomic datasets, built over decades, revealed why females are less frequently diagnosed than males. Females—especially those with cognitive and motor challenges—carry a higher burden of de novo variants than males, despite no difference in the specific genes affected.15 This pattern supports the concept of differential liability, in which females may have greater biological resilience to autism-related risk.
Additional findings suggest that sex differences arise from multiple mechanisms beyond a single female protective effect. Studies showed that female siblings of individuals with autism have language difficulties that are milder than those seen in autism but greater than those in unrelated peers.16 Other work found that although females are typically diagnosed later overall17, among children diagnosed before age two, females outnumber males.18 Females also have higher rates of co-occurring mental health conditions, which may complicate or delay diagnosis.18 These differences may reflect both biological variation and diagnostic bias, which could be addressed through clinician training and refinement of diagnostic instruments.19 For example, Black autistic girls are less likely to receive a diagnosis despite similar social responsiveness scores, underscoring the role of bias in diagnostic practices.20
Other biological mechanisms underlying sex differences involve the X chromosome. Because females have two X chromosomes, understanding X-linked gene expression in both sexes is critical. Research in 2025 identified 33 X-linked variants consistently associated with autism.21 These variants are involved in brain development at different stages and show sex-specific expression patterns.21 Differences between X- and Y-linked genes may further contribute to sex differences in autism presentation and diagnosis.
Moving Toward Precision Medicine
In 2025, multiple studies used organoids and genetic medicines to advance targeted, personalized approaches for autism, particularly in individuals with known genetic conditions. Organoid and assembloid technologies allow scientists to observe early brain development and identify where developmental pathways diverge in autism. Organoids derived from an individual’s own cells can model that person’s unique neurobiology, enabling researchers to test how brain circuits develop and respond to interventions in a highly personalized way. For individuals with autism, this approach may help identify treatments tailored to specific genetic and cellular profiles rather than relying on one-size-fits-all strategies.
Organoids are being used to develop personalized interventions for conditions including FMR1-related disorders, Timothy syndrome, MECP2-related disorders, and Dup15q syndrome.22,23 These studies include efforts to predict responses to anti-seizure medications24 and to better understand early cellular processes that influence autism risk.25
Similarly, researchers made progress in genetic medicines, with early studies demonstrating the feasibility for autism linked to highly penetrant genetic variants. Approaches using antisense oligonucleotides (ASOs), CRISPR-based tools, and RNA repair strategies have advanced from animal models toward human trials, including clinical studies in Angelman syndrome26 and Rett syndrome.27 These advances are moving the field from gene discovery toward functional recovery.28 Additional progress has brought other potential therapeutics closer to autism-specific trials.28 While gene therapies target specific mutations, treatments developed for one rare genetic condition are now being explored in related conditions and, potentially, in idiopathic autism. Examples include trials of IGF-129,30 and metformin.31 Developing an evidence base for cross-disorder treatments will open the door to further expanding their utility in autism without an established genetic cause.
Predicting the Future Using a Longitudinal Design
Families often wonder how their child with autism will develop throughout their life and how they should prepare.32 Studying individuals over time, rather than at a single age, has revealed important insights into outcomes, including which features are likely to remain stable and which may improve. In general, individuals with lower baseline abilities tend to show more challenging developmental trajectories, although this pattern is not universal.33,34 Environmental factors, such as socioeconomic status, may also influence both baseline abilities and developmental trajectories.33 Cognitive ability was the most predictive of core autism symptom trajectory, indicating it strongly predicts ASD outcomes.
At a neurobiological level, developmental trajectories may be partially explained by autism-related differences in temporal lobe white matter development. Autistic brains show altered patterns of synaptic pruning during development, which may affect how neural circuits mature over time.35
Research using rare and critical postmortem brain tissue from individuals with autism has begun to show how the autistic brain changes across the lifespan at a cellular level. However, this line of research is severely limited by the scarcity of available brain tissue. Future studies should examine distinct autism subtypes or subgroups—defined by behavioral features or co-occurring conditions—to better understand differences in brain development. Funding agencies should recognize the long-term value of longitudinal and tissue-based research and support sustained staffing, participant engagement, and the evolving needs of families followed over time.32
Early Intervention Promotes Improvement
This year, scientists compiled large datasets to study predictors of early intervention outcomes. These studies examined a range of intervention modalities targeting core autism features. Researchers found that intervention effectiveness is influenced by factors such as duration and intensity,36,37 baseline skill levels at the start of intervention,36–38 and earlier age at entry,37,38 but not by the specific name or branded model of the intervention. In other words, interventions with different names that focus on skill learning and the promotion of social and cognitive development were broadly beneficial.
By contrast, duration, intensity, and earlier age at entry were consistently associated with improvements in cognitive and language abilities.36,37 Because young children receive much of their social interaction from parents or other caregivers, parent involvement has proven to be a critical component of effective early intervention.39 These approaches are now being applied successfully to infants who show early signs of developmental challenges, extending intervention efforts to even earlier stages of development.40
Managing Severe, Intense, and Dangerous Behaviors
Meaningful progress was also made this year in understanding severe, intense, and dangerous behaviors in autism. This included improved understanding of wandering, a behavior associated with increased risk of injury and death, and more common among autistic children than their non-autistic peers (DiGiuseppi), as well as the development of more effective interventions to reduce wandering (Scheithauer).
This year also saw increased research attention to catatonia, a potentially fatal condition that is more prevalent among individuals with autism and intellectual and developmental disabilities, yet often more difficult to recognize in autistic individuals than in those without an autism diagnosis (Smith). Cases of documented catatonia occur in about 10% of people with autism, although this might be an underestimation due to communication difficulties in individuals with autism, or changes in development over time that may be mistaken for a core autism feature. You will be hearing more about this issue from ASF in the future.
Conclusion
Despite political hostility and the spread of misinformation that disrupted some lines of research, autism scientists rose to the occasion in 2025. They shared discoveries that help families better understand the biological diversity of autism and the nature of different autism subtypes. These findings illustrate both the neurobiology underlying distinct forms of autism and the role environmental factors play in shaping outcomes. Research in 2025 also highlighted both the overlap and the unique features of autism compared with other neurodevelopmental conditions, such as ADHD. Together, these advances move the field closer to ensuring that the right person receives the right treatment at the right time.
References
- Litman A, Sauerwald N, Green Snyder L, et al. Decomposition of phenotypic heterogeneity in autism reveals underlying genetic programs. Nat Genet. 2025;57(7):1611-1619. doi:10.1038/s41588-025-02224-z
- Zhang X, Grove J, Gu Y, et al. Polygenic and developmental profiles of autism differ by age at diagnosis. Nature. 2025;646(8087):1146-1155. doi:10.1038/s41586-025-09542-6
- Kim SW, Lee H, Song DY, et al. Evaluation of familial phenotype deviation to measure the impact of de novo mutations in autism. Genome Med. 2025;17(1):93. doi:10.1186/s13073-025-01532-7
- Segura P, Pagani M, Bishop SL, et al. Connectome-based symptom mapping and in silico related gene expression in children with autism and/or attention-deficit/hyperactivity disorder. Mol Psychiatry. Published online October 23, 2025. doi:10.1038/s41380-025-03205-8
- Grotzinger AD, Werme J, Peyrot WJ, et al. Mapping the genetic landscape across 14 psychiatric disorders. Nature. Published online December 10, 2025. doi:10.1038/s41586-025-09820-3
- Pecci-Terroba C, Lai MC, Lombardo MV, et al. Subgrouping autism and ADHD based on structural MRI population modelling centiles. Mol Autism. 2025;16(1):33. doi:10.1186/s13229-025-00667-z
- Seelemeyer H, Gurr C, Leyhausen J, et al. Decomposing the Brain in Autism: Linking Behavioral Domains to Neuroanatomical Variation and Genomic Underpinnings. Biol Psychiatry Cogn Neurosci Neuroimaging. 2025;10(10):1067-1077. doi:10.1016/j.bpsc.2024.12.003
- Mailick M, Bennett T, DaWalt LS, et al. Expanding Research on Contextual Factors in Autism Research: What Took Us So Long? Autism Res Off J Int Soc Autism Res. 2025;18(4):710-716. doi:10.1002/aur.3312
- Zhang Y, Yahia A, Sandin S, Åden U, Tammimies K. Prematurity and genetic liability for autism spectrum disorder. Genome Med. 2025;17(1):108. doi:10.1186/s13073-025-01552-3
- Chatzigeorgiou C, Asgel Z, Avila MN, et al. Autism Heterogeneity Related to Preterm Birth: Multi-Ancestry Results From the Simons Foundation Powering Autism Research for Knowledge Sample. Biol Psychiatry Glob Open Sci. 2026;6(1):100614. doi:10.1016/j.bpsgos.2025.100614
- Isaksson J, Eklund F, Remnélius KL, Black MH, Bölte S. Neurodevelopmental conditions and adaptive functioning – a co-twin control study. J Child Psychol Psychiatry. n/a(n/a). doi:https://doi.org/10.1111/jcpp.70073
- Key AP, Jones D, Corbett BA. Social Functioning in Autistic Children with Below-Average vs. Average IQ: Limited Behavioral and Neural Evidence of Group Differences. J Autism Dev Disord. Published online March 13, 2025. doi:10.1007/s10803-025-06755-6
- Salenius K, Väljä N, Thusberg S, et al. Exploring autism spectrum disorder and co-occurring trait associations to elucidate multivariate genetic mechanisms and insights. BMC Psychiatry. 2024;24(1):934. doi:10.1186/s12888-024-06392-w
- Lee BK, Stephansson O, Gardner RM. Paracetamol (acetaminophen) use in pregnancy and risk of autism and ADHD. BMJ. 2025;391:r2438. doi:10.1136/bmj.r2438
- Koko M, Satterstrom FK, Autism Sequencing Consortium, APEX consortium, Warrier V, Martin H. Contribution of autosomal rare and de novo variants to sex differences in autism. Am J Hum Genet. 2025;112(3):599-614. doi:10.1016/j.ajhg.2025.01.016
- Belenger M, Dumont C, Kissine M. Exploring Pragmatic Abilities in Sisters of Autistic Individuals: A Methodological Solution to Female Autism Research. Autism Res Off J Int Soc Autism Res. Published online November 18, 2025. doi:10.1002/aur.70147
- Maciver D, Singh Roy A, Johnston L, et al. Are we getting better at identifying and diagnosing neurodivergent girls and women? Insights into sex ratios and age of diagnosis from clinical population data in Scotland. Autism Int J Res Pract. Published online October 28, 2025:13623613251383343. doi:10.1177/13623613251383343
- Chen YJ, Lai MC, Georgiades S, et al. Initial diagnosis patterns of coexisting mental health and neurodevelopmental conditions in autistic children and youth: Evidence from a nationally representative sample in Canada. J Child Psychol Psychiatry. Published online September 1, 2025. doi:10.1111/jcpp.70039
- Burrows CA, Sung S, Zheng S, et al. Sex-Related Measurement Bias in Autism Spectrum Disorder Symptoms in the Baby Siblings Research Consortium. JAMA Netw Open. 2025;8(8):e2525887. doi:10.1001/jamanetworkopen.2025.25887
- Lyall K, Dickerson AS, Green AM, et al. Demographic Correlates of Autism: How Do Associations Compare Between Diagnosis and a Quantitative Trait Measure? Autism Res Off J Int Soc Autism Res. 2025;18(3):648-659. doi:10.1002/aur.3296
- Mendes M, Chen DZ, Engchuan W, et al. Chromosome X-wide common variant association study in autism spectrum disorder. Am J Hum Genet. 2025;112(1):135-153. doi:10.1016/j.ajhg.2024.11.008
- Michels S, Mali A, Jäntti H, Rezaie M, Malm T. Microglial involvement in autism spectrum disorder: insights from human data and iPSC models. Brain Behav Immun. 2025;130:106071. doi:10.1016/j.bbi.2025.106071
- Perez Y, Velmeshev D, Wang L, et al. Single-cell analysis of dup15q syndrome reveals developmental and postnatal molecular changes in autism. Nat Commun. 2025;16(1):6177. doi:10.1038/s41467-025-61184-4
- Yang Y, Cai Y, Wang S, et al. Human Cortical Organoids with a Novel SCN2A Variant Exhibit Hyperexcitability and Differential Responses to Anti-Seizure Compounds. Neurosci Bull. 2025;41(11):2010-2024. doi:10.1007/s12264-025-01429-w
- Stankovic I, Smit P, Cross J, et al. Extracellular vesicle profiling reveals novel autism signatures in patient-derived forebrain organoids. Transl Psychiatry. 2025;15(1):393. doi:10.1038/s41398-025-03607-w
- Hipp JF, Bacino CA, Bird LM, et al. The UBE3A-ATS antisense oligonucleotide rugonersen in children with Angelman syndrome: a phase 1 trial. Nat Med. 2025;31(9):2936-2945. doi:10.1038/s41591-025-03784-7
- Ribeiro FCP, Alves ML, Meneses AC, et al. Mecasermin for the treatment of Rett Syndrome: a systematic review. Neurogenetics. 2025;26(1):78. doi:10.1007/s10048-025-00860-5
- Devinsky O, Coller J, Ahrens-Nicklas R, et al. Gene therapies for neurogenetic disorders. Trends Mol Med. 2025;31(9):814-826. doi:10.1016/j.molmed.2025.01.015
- Aria F, Arp CJ, Prikas E, et al. A prodrug targeting CIM6P/IGF2R enhances memory in healthy mice and reverses deficits in an Angelman syndrome mouse model. Transl Psychiatry. 2025;15(1):438. doi:10.1038/s41398-025-03610-1
- Percy AK, Ryther R, Marsh ED, et al. Results from the phase 2/3 DAFFODIL study of trofinetide in girls aged 2-4 years with Rett syndrome. Med N Y N. 2025;6(6):100608. doi:10.1016/j.medj.2025.100608
- Zhu Y, Li D, Hu C, et al. Effects of Metformin on children with Fragile X Syndrome: a randomized, double-blind, placebo-controlled trial. Mol Autism. 2025;16(1):57. doi:10.1186/s13229-025-00691-z
- Hus Bal V, Szatmari P, Georgiades S, et al. Methodological challenges and opportunities when studying the course of autism. Autism Int J Res Pract. 2025;29(10):2618-2631. doi:10.1177/13623613251341012
- Hiralal KR, Dieleman GC, Kok BR, et al. Latent trajectories in autistic individuals: A systematic review. Autism. Published online September 25, 2025:13623613251370818. doi:10.1177/13623613251370818
- S Dichter G, Grosser KF, Cummings KK, et al. Changes in Autism Traits from Early Childhood To Adolescence in the Study To Explore Early Development. J Autism Dev Disord. Published online November 8, 2025. doi:10.1007/s10803-025-07107-0
- Hanson KL, Avino T, Taylor SL, Murray KD, Schumann CM. Age-related differences in axon pruning and myelination may alter neural signaling in autism spectrum disorder. Mol Autism. 2025;16(1):53. doi:10.1186/s13229-025-00684-y
- Chetcuti L, Uljarević M, Schuck RK, et al. Characterizing predictors of response to behavioral interventions for children with autism spectrum disorder: A meta-analytic approach. Clin Psychol Rev. 2025;119:102588. doi:10.1016/j.cpr.2025.102588
- Vivanti G, Lombardo MV, Zitter A, et al. Proportion and Profile of Autistic Children Not Acquiring Spoken Language Despite Receiving Evidence-Based Early Interventions. J Clin Child Adolesc Psychol Off J Soc Clin Child Adolesc Psychol Am Psychol Assoc Div 53. Published online November 20, 2025:1-18. doi:10.1080/15374416.2025.2579286
- Mandelli V, Busuoli EM, Godel M, et al. Mega-analytic support for Early Start Denver Model, age at intervention start, and pre-intervention developmental level as factors differentiating early intervention outcomes in autism. MedRxiv Prepr Serv Health Sci. Published online April 16, 2025:2025.04.14.25325786. doi:10.1101/2025.04.14.25325786
- Kuhn J, Menon N, Nunez-Pepen R, et al. Parent Use and Perceptions of Problem-Solving Education in the Context of Parent-Implemented Intervention for Toddlers With Early Signs of Autism. J Autism Dev Disord. Published online November 6, 2025. doi:10.1007/s10803-025-07071-9
- Dowds E, MacWilliam S, Solish A, et al. A pilot evaluation of the Baby Social ABCs caregiver-mediated intervention for 6-15-month-olds with early signs of autism-feasibility, acceptability, and preliminary evidence. Front Child Adolesc Psychiatry. 2025;4:1689781. doi:10.3389/frcha.2025.1689781
- DiGuiseppi C, Holst B, Bolt M, et al. Associations of Adaptive Behavior and Wandering with Serious Injuries in Young Children with Autism: Study to Explore Early Development. J Autism Dev Disord. Published online August 20, 2025. doi:10.1007/s10803-025-06978-7
- Scheithauer M, Lomas Mevers J, Scahill L, et al. A Randomized Trial of Caregiver-Mediated Function-Based Elopement Treatment for Autistic Children. Autism Int J Res Pract. 2025;29(8):1973-1986. doi:10.1177/1362361325133038843. Smith JR, Lim S, Bindra S, et al. Longitudinal Symptom Burden and Pharmacologic Management of Catatonia in Autism With Intellectual Disability: An Observational Study. Autism Res Off J Int Soc Autism Res. 2025;18(2):449-462. doi:10.1002/aur.3315
The post 2025 Autism Research Year in Review appeared first on Autism Science Foundation.
]]>The post Take Our “Effect of Funding Loss” Survey appeared first on Autism Science Foundation.
]]>The post Take Our “Effect of Funding Loss” Survey appeared first on Autism Science Foundation.
]]>The post Reaffirming our Commitment: A Message from Our Founder appeared first on Autism Science Foundation.
]]>This journey has not been without obstacles. One of the greatest challenges ASF has faced—and continues to confront—is the spread of misinformation that falsely links vaccines to autism. We have remained steadfast in defending science, countering dangerous myths, and ensuring that facts guide public understanding. Today, our resolve remains stronger than ever.
Despite uncertainty in research funding and shifting government priorities, ASF’s mission does not waver. We are resolute in our commitment to advance research that deepens understanding of the causes of autism and leads to meaningful treatments that improve lives.
As we navigate this evolving landscape, we do so with a clear objective: to push the boundaries of research, to support scientists seeking answers, and to prioritize the needs of the autism community. We invite you to join us for this next chapter of the journey.
In these critical times, ASF will continue to rely on our dedicated supporters—you are essential to sustaining the progress we have made. Together, we will ensure that research moves forward, guided by science, driven by purpose, and rooted in a commitment to our loved ones with autism.
With gratitude,
Alison Singer
President, Autism Science Foundation
The post Reaffirming our Commitment: A Message from Our Founder appeared first on Autism Science Foundation.
]]>The post RFA for 2025 Undergrad Summer Research Now Available appeared first on Autism Science Foundation.
]]>The post RFA for 2025 Undergrad Summer Research Now Available appeared first on Autism Science Foundation.
]]>The post 2024 Autism Science Review appeared first on Autism Science Foundation.
]]>This year’s progress in autism research includes promising findings, clarifications, explanations, and the uncovering of new avenues of inquiry. The focus is now on personalized medicine: finding the right treatment for the right person at the right time through targeted interventions. Advances in technology and genetic testing are opening new avenues for therapeutics, rapid drug testing, and improved differentiation of subgroups of autism.
Listen to the 2024 Year End Summary here.
The Value of Genetic Testing
With the improvement of genetic testing techniques, identifying genes contributing to an autism diagnosis is becoming increasingly precise. Currently, a genetic cause of autism can be pinpointed in around 20% of cases (1). While guidelines vary slightly, most professional organizations associated with autism recommend genetic testing for affected families (2). Testing can aid our understanding of autism symptoms as well as provides therapeutic options for individuals with a diagnosis (3). One example of progress in autism treatment involves Rett Syndrome, a developmental disorder that includes many autistic features. Last year, the FDA approved the drug trofinetide for Rett Syndrome, which affects growth factor signaling. This year, the long-term efficacy of trofinetide was demonstrated (4).

A promising new tool revealed this year is the use of brain organoids to identify and develop more personalized gene therapies for individuals with rare genetic syndromes. For instance, Timothy Syndrome is characterized by autism and heartbeat irregularities. Utilizing cells derived from individuals with Timothy Syndrome, researchers in the Pasca lab at Stanford University developed a targeted gene therapy that corrected deficits in cell migration and electrical signaling—a significant step toward personalized clinical therapy (5). This advancement provides hope not just for Timothy Syndrome but for many rare genetic disorders associated with autism. Although these genetic variants are rare, therapies developed for specific cases have the potential for broader applications.
In addition to using derived brain organoids, which are grown in a dish, scientists are also studying the actual brain tissue of deceased individuals with autism. For example, the Geschwind Lab at UCLA, via the Autism BrainNet program, has studied gene activities directly in the brain. They found that genes controlling neuronal function, migration, cell-to-cell signaling, and neuronal communication are downregulated, while those controlling immune system activation are upregulated (6). These findings, initially observed in blood samples, have now been validated in brain tissue. Researchers have also identified changes in gene expression across different brain cell types organized into distinct networks (7). These discoveries show that, even after decades of research, new basic science findings continue to reshape our understanding of autism (8).
Advances in Measuring Outcomes
Progress was made this year in addressing a persistent challenge in behavioral studies: how to measure outcomes effectively. A new tool, the Brief Observation of Social Communication Change (BOSCC), was developed to assess the efficacy of different therapies, moving beyond traditional diagnostic measures that were not designed for this purpose (9). The BOSCC has been used across various studies to demonstrate the benefits of early intervention (9). Remote assessments, adapted during COVID-19, also showed validity in measuring social attention in rare genetic syndromes associated with autism, with potential for broader application (10-12).
Research also focused on utilizing existing instruments to measure clinically-significant change, bridging the gap between laboratory results and real-life impact. While not as high-profile as medication or therapy, these methodological advances are crucial for ensuring the success of treatment trials (13,14).
Environmental Factors: What Remains to Be Explored?
Understanding which environmental factors contribute to autism remains a formidable challenge. This year, several studies provided new data, clarified previous findings, and examined novel factors. For instance, while prenatal infections and immune challenges are still considered risk factors for autism, additional studies suggest that these relationships may be moderated by genetics or shared familial factors (15,16). Concerns about COVID-19 infection during pregnancy being linked to an autism diagnosis inn those children were alleviated by two studies showing no association between maternal COVID-19 and autism in offspring (17). Similarly, neither cannabis use during pregnancy (18) nor prenatal diet (19) was linked to an autism diagnosis. Increased screen time was associated with preschool autism traits and lower developmental scores (20,21), furthering the long-held belief that multiple genetic and environmental factors may contribute to an autism diagnosis rather than one single factor being the cause.
One potential mechanism linking environmental factors such as immune challenges or antiepileptic drugs to autism risk involves the timing of brain cell activation during early development (22). Disruptions in these processes may hinder proper brain development, a theory increasingly supported by research into neurodevelopmental disorders.
Recurrence Rates
Decades of evidence show that individuals with a family history of autism are at higher risk of diagnosis compared to the general population. High-risk groups include those born prematurely (23) or with rare genetic disorders associated with autism. The Baby Siblings Research Consortium (BSRC) updated recurrence risk statistics for families with one autistic child. While the general population risk is 1 in 36, younger siblings face a risk of 1 in 5 (24). Following children to school age, the numbers are higher (25). Furthermore, the risk increases for families with multiple autistic children in the family. Interestingly, recurrence risk is higher when the older sibling is female, suggesting a protective effect in some females that may be outweighed by genetic liability in others (24,26).
Additional data from Denmark’s health registries revealed that individuals with extended family histories of autism or psychiatric disorders, such as bipolar disorder and schizophrenia, have an increased likelihood of autism diagnoses (27). Longitudinal studies, like the Autism Science Foundation’s Next Gen Sibs Project, continue to explore these familial relationships over time.

The Enduring Question: How Many People Have Autism?
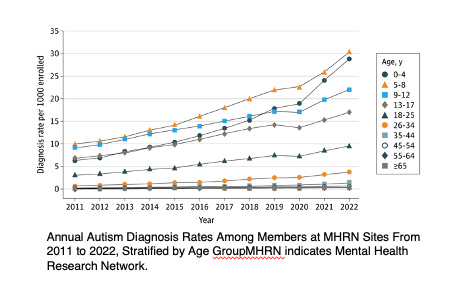
Each year, families, researchers, and public health officials ask, “How many people have autism?” The CDC’s Autism and Developmental Disabilities Monitoring (ADDM) Network estimates the prevalence every two years. The latest report calculated a rate of 1 in 36 for eight-year-olds. This year, a study using a large healthcare database confirmed rising prevalence rates, an increase in adult diagnoses, and a growing recognition of autism in females, although females remain underdiagnosed compared to males (28,29). Late-diagnosed individuals often exhibit a different autism phenotype, characterized by symptoms that allow for more social camouflaging (30,31).
The Spectrum Within Autism
Autism manifests in a wide range of presentations. Some individuals, described as having “profound autism,” exhibit severe intellectual disabilities, limited language, and require constant supervision. These individuals also display higher rates of aggression, self-injury, and stereotyped behaviors, often due to their inability to communicate distress (32-36). Research increasingly shows that biologically, individuals with profound autism differ significantly from those with non-profound autism. For example, brain imaging studies reveal that individuals with profound autism exhibit distinct brain structures and functions, including widespread brain overgrowth (37,38). The differentiation between non-profound and profound autism can sometimes be made quickly (39,40) and is based on features like adaptive abilities, motor skills, language, and cognitive abilities (38).
Families of individuals with profound autism face unique challenges related to stigma, accommodation, and access to support. Despite progress in public awareness, societal perceptions often marginalize those with severe disabilities. Experts like Drexel University’s Dr. Giacomo Vivanti advocate for embracing the complexity and nuance of autism, acknowledging that the experiences of those with profound autism differ substantially from others on the spectrum (41).

Insights into Language Development
Severe language deficits are common in individuals with profound autism, but language and communication challenges are widespread across the spectrum. While language impairments are no longer part of the DSM5 criteria, they are critical in understanding the nature of autism. This year, research identified genetic links, such as neurexin family genes, that affect language abilities (42). Studies also showed that minimally verbal individuals often have better receptive language skills than expressive skills, emphasizing the importance of tailored language development strategies (43). Parent language input and social skills also play critical roles in shaping language outcomes (44).
Gender Differences in Autism

Large studies highlight significant gender differences in autism. Males show higher heritability rates and are more frequently diagnosed, while females with autism are more likely to have intellectual disabilities (26). Biological differences may contribute to these disparities, including gene expression and neural connectivity (45-48). Understanding these differences is crucial for addressing issues such as the higher rates of suicide and anxiety observed in autistic females (49-51).
References:
- Wright JR, Astrovskaya I, Barns SD, et al. Return of genetic research results in 21,532 individuals with autism. Genet Med 2024; 26(10): 101202.
- Srivastava S, Cole JJ, Cohen JS, et al. Survey of the Landscape of Society Practice Guidelines for Genetic Testing of Neurodevelopmental Disorders. Ann Neurol 2024; 96(5): 900-13.
- Leblond CS, Rolland T, Barthome E, et al. A Genetic Bridge Between Medicine and Neurodiversity for Autism. Annu Rev Genet 2024; 58(1): 487-512.
- Percy AK, Neul JL, Benke TA, et al. Trofinetide for the treatment of Rett syndrome: Long-term safety and efficacy results of the 32-month, open-label LILAC-2 study. Med 2024; 5(10): 1275-81 e2.
- Chen X, Birey F, Li MY, et al. Antisense oligonucleotide therapeutic approach for Timothy syndrome. Nature 2024; 628(8009): 818-25.
- Xia Y, Xia C, Jiang Y, et al. Transcriptomic sex differences in postmortem brain samples from patients with psychiatric disorders. Sci Transl Med 2024; 16(749): eadh9974.
- Wamsley B, Bicks L, Cheng Y, et al. Molecular cascades and cell type-specific signatures in ASD revealed by single-cell genomics. Science 2024; 384(6698): eadh2602.
- Tranfaglia M. In: Halladay A, editor. email; 2024.
- Swain D, Li Y, Brown HR, et al. Implementing a Uniform Outcome Measurement Approach for Early Interventions of Autism Spectrum Disorders. J Am Acad Child Adolesc Psychiatry 2024.
- Frazier TW, Busch RM, Klaas P, et al. Quantifying neurobehavioral profiles across neurodevelopmental genetic syndromes and idiopathic neurodevelopmental disorders. Dev Med Child Neurol 2024.
- Frazier TW, Busch RM, Klaas P, et al. Remote monitoring of social attention in neurogenetic syndromes and idiopathic neurodevelopmental disability. Autism Res 2024.
- Nicholas DB, Zulla RT, Cielsielski J, Zwaigenbaum L, Conlon O. COVID-19 Pandemic Experiences of Families in Which a Child/Youth Has Autism and Their Service Providers: Perspectives and Lessons Learned. J Autism Dev Disord 2024.
- Schiltz HK, Williams ZJ, Zheng S, et al. Measurement matters: A commentary on the state of the science on patient reported outcome measures (PROMs) in autism research. Autism Res 2024; 17(4): 690-701.
- Nelson MA, Schmitt LM, Horn PS, et al. Parent-Reported Outcome Measures for Individuals with Fragile X Syndrome: Clinically Meaningful Change Thresholds. J Autism Dev Disord 2024.
- Gardner RM, Brynge M, Sjoqvist H, Dalman C, Karlsson H. Maternal immune activation and autism in the offspring-what is the evidence for causation? Biol Psychiatry 2024.
- Madley-Dowd P, Ahlqvist VH, Forbes H, et al. Antiseizure medication use during pregnancy and children’s neurodevelopmental outcomes. Nat Commun 2024; 15(1): 9640.
- Firestein MR, Manessis A, Warmingham JM, et al. Positive Autism Screening Rates in Toddlers Born During the COVID-19 Pandemic. JAMA Netw Open 2024; 7(9): e2435005.
- Avalos LA, Shenkute M, Alexeeff SE, et al. Maternal Prenatal Cannabis Use and Child Autism Spectrum Disorder. JAMA Netw Open 2024; 7(10): e2440301.
- Vecchione R, Westlake M, Bragg MG, et al. Maternal Dietary Patterns During Pregnancy and Child Autism-Related Traits in the Environmental Influences on Child Health Outcomes Consortium. Nutrients 2024; 16(22).
- Hill MM, Gangi DN, Miller M. Toddler Screen Time: Longitudinal Associations with Autism and ADHD Symptoms and Developmental Outcomes. Child Psychiatry Hum Dev 2024.
- Lin PI, Wu WT, Guo YL. Screen Time Before 2 Years of Age and Risk of Autism at 12 Years of Age. JAMA Pediatr 2024.
- Godavarthi SK, Li HQ, Pratelli M, Spitzer NC. Embryonic exposure to environmental factors drives transmitter switching in the neonatal mouse cortex causing autistic-like adult behavior. Proc Natl Acad Sci U S A 2024; 121(35): e2406928121.
- Hamner T, Perez Liz G, Kelly K, et al. Autism screening and diagnostic outcomes among toddlers born preterm. Dev Med Child Neurol 2024.
- Ozonoff S, Young GS, Bradshaw J, et al. Familial Recurrence of Autism: Updates From the Baby Siblings Research Consortium. Pediatrics 2024; 154(2).
- Bazelmans T, Arthur R, Pasco G, et al. Mid-childhood autism sibling recurrence in infants with a family history of autism. Autism Res 2024; 17(7): 1501-14.
- Kim SW, Lee H, Song DY, et al. Whole genome sequencing analysis identifies sex differences of familial pattern contributing to phenotypic diversity in autism. Genome Med 2024; 16(1): 114.
- Schendel D, Ejlskov L, Overgaard M, et al. 3-generation family histories of mental, neurologic, cardiometabolic, birth defect, asthma, allergy, and autoimmune conditions associated with autism: An open-source catalog of findings. Autism Res 2024; 17(10): 2144-55.
- Grosvenor LP, Croen LA, Lynch FL, et al. Autism Diagnosis Among US Children and Adults, 2011-2022. JAMA Netw Open 2024; 7(10): e2442218.
- Harrop C, Tomaszewski B, Putnam O, Klein C, Lamarche E, Klinger L. Are the diagnostic rates of autistic females increasing? An examination of state-wide trends. J Child Psychol Psychiatry 2024; 65(7): 973-83.
- Russell AS, McFayden TC, McAllister M, et al. Who, when, where, and why: A systematic review of “late diagnosis” in autism. Autism Res 2024.
- Sturm A, Huang S, Bal V, Schwartzman B. Psychometric exploration of the RAADS-R with autistic adults: Implications for research and clinical practice. Autism 2024; 28(9): 2334-45.
- Ferguson EF, Barnett ML, Goodwin JW, Vernon TW. “There is No Help:” Caregiver Perspectives on Service Needs for Adolescents and Adults with Profound Autism. J Autism Dev Disord 2024.
- Ferguson EF, Spackman E, Cai RY, Hardan AY, Uljarevic M. Characterizing associations between emotion dysregulation, anxiety, and repetitive behaviors in autistic youth with intellectual disability. Autism Res 2024; 17(9): 1810-7.
- Gohari D, Schiltz H, Lord C. A Longitudinal Study of Aggression in People with Autism and Other Neurodevelopmental Disabilities. J Autism Dev Disord 2024.
- Clarke EB, McCauley JB, Lutz A, Gotelli M, Sheinkopf SJ, Lord C. Understanding profound autism: implications for stigma and supports. Front Psychiatry 2024; 15: 1287096.
- Romani PW, Luehring MC, Koerner AM, Baikie SM. Characterization and Outcomes of Profoundly and Non-profoundly Autistic Individuals Admitted to aSpecialized Psychiatric Inpatient Unit. J Autism Dev Disord 2024.
- Courchesne E, Taluja V, Nazari S, et al. Embryonic origin of two ASD subtypes of social symptom severity: the larger the brain cortical organoid size, the more severe the social symptoms. Mol Autism 2024; 15(1): 22.
- Mandelli V, Severino I, Eyler L, Pierce K, Courchesne E, Lombardo MV. A 3D approach to understanding heterogeneity in early developing autisms. Molecular Autism 2024; 15(1): 41.
- Canale RR, Larson C, Thomas RP, Barton M, Fein D, Eigsti IM. Investigating frank autism: clinician initial impressions and autism characteristics. Mol Autism 2024; 15(1): 48.
- Sterrett K, Clarke E, Nofer J, Piven J, Lord C. Toward a functional classification for autism in adulthood. Autism Res 2024; 17(10): 2105-19.
- Vivanti G. Autism Early Intervention – Progress, Steps Backward, and the Reconciliation of Conflicting Narratives. Curr Psychiatry Rep 2024.
- Arutiunian V, Santhosh M, Neuhaus E, et al. A common genetic variant in the Neurexin family member CNTNAP2 is related to language but not communication skills in youth with Autism Spectrum Disorder. Autism Res 2024.
- Pizzano M, Shire S, Shih W, et al. Profiles of minimally verbal autistic children: Illuminating the neglected end of the spectrum. Autism Res 2024; 17(6): 1218-29.
- Pecukonis M, Butler LK, Tager-Flusberg H. What role does the environment play in language development? Exploring the associations among socioeconomic status, parent language input, and language skills in school-aged children with autism. Autism Res 2024.
- Kissel LT, Pochareddy S, An JY, et al. Sex-Differential Gene Expression in Developing Human Cortex and Its Intersection With Autism Risk Pathways. Biol Psychiatry Glob Open Sci 2024; 4(4): 100321.
- Del Bianco T, Lai MC, Mason L, et al. Sex differences in social brain neural responses in autism: temporal profiles of configural face-processing within data-driven time windows. Sci Rep 2024; 14(1): 14038.
- Khan YT, Tsompanidis A, Radecki MA, et al. Sex Differences in Human Brain Structure at Birth. Biol Sex Differ 2024; 15(1): 81.
- Li H, Zhang Q, Duan T, et al. Sex differences in brain functional specialization and interhemispheric cooperation among children with autism spectrum disorders. Sci Rep 2024; 14(1): 22096.
- Hull L, Stark I, Lundberg M, et al. Sex differences in self-harm and suicide in young autistic adults. Acta Psychiatr Scand 2024; 150(4): 223-33.
- Long EE, Johnson MF, Carpenter LA. Autistic Characteristics, Cognitive Impairment, and Sex as Predictors of Anxiety and Depression among Autistic Youth. J Autism Dev Disord 2024.
- Santomauro DF, Hedley D, Sahin E, et al. The global burden of suicide mortality among people on the autism spectrum: A systematic review, meta-analysis, and extension of estimates from the Global Burden of Disease Study 2021. Psychiatry Res 2024; 341: 116150.
The post 2024 Autism Science Review appeared first on Autism Science Foundation.
]]>The post Top 10 2024 Publications From ASF-Sponsored Research appeared first on Autism Science Foundation.
]]>Familial Recurrence of Autism: Updates From the Baby Siblings Research Consortium
PubMed ID: 39011552
#2 Many autism-related genes work through a druggable pathway called mTOR.
Dysregulation of mTOR signaling mediates common neurite and migration defects in both idiopathic and 16p11.2 deletion autism neural precursor cells
PubMed ID: 38525876
#3 One of the most effective methods for improving social communication and peer interactions in children is adapted for adults.
Adapting the PEERS® for Young Adults Program for Autistic Adults across the Lifespan
PubMed ID: 39201145
#4 A new intervention improves mental health in adolescents and young adults with ASD.
Results from a Pilot Randomized Controlled Trial of a Single-Session Growth-Mindset Intervention for Internalizing Symptoms in Autistic Youth
PubMed ID: 38833031 .
#5 A new online version of a tool to measure cognitive ability is a promising alternative to in-person assessments for children and adolescents with ASD.
An Exploration of Online and In-Person Administration of the Kaufman Brief Intelligence Test, Second Edition (KBIT-2) in Children and Adolescents Being Evaluated for Autism Spectrum Disorder
PubMed ID: 38536637
#6 The impact of gastrointestinal issues on behavioral challenges and quality of life in those with profound autism is shared directly with GI doctors.
Conference proceedings: Inaugural meeting of the consortium for autism, genetic neurodevelopmental disorders, and digestive diseases
PubMed ID: 39257288
#7 A short screening tool can identify profound autism in adults.
Toward a functional classification for autism in adulthood
PubMed ID: 39031157
#8 Families of people with profound autism describe specific and unique challenges across their lifespan.
Profound Autism: An Imperative Diagnosis
PubMed ID: 38423722
#9 Teachers may overlook autistic school- age females
Sex Differences and Parent–Teacher Discrepancies in Reports of Autism Traits: Evidence for Camouflaging in a School Setting
PubMed ID: 39060706
#10 Conducting studies in schools reduces racial/ethnic disparities in research.
Examining race, ethnicity, sex, and gender among autistic youth and their educators who participated in school-based research
PubMed ID: 39056323
The post Top 10 2024 Publications From ASF-Sponsored Research appeared first on Autism Science Foundation.
]]>The post Day of Learning Tickets Now Available appeared first on Autism Science Foundation.
]]>The post Day of Learning Tickets Now Available appeared first on Autism Science Foundation.
]]>The post 2023 Year End Review appeared first on Autism Science Foundation.
]]>What is autism?
This year the community had a lot of conversations on how autism should be defined and diagnosed.
Autism’s heterogeneity complicates issues relating to research, advocacy, services, supports and medical care. Some experience autism as a debilitating disorder, while others consider it an identity. Many fall in the middle. In 2021, the term “profound autism” was introduced to describe autistic individuals who require 24/7 supports, who are minimally verbal or nonspeaking or who have an IQ under 50. This year, there were several papers that examined this new definition and two editorials that attempted to reconcile the different views around the broad autism spectrum.1,2 Greene and Whitehouse suggest that individual brain differences, originating genetically or as the result of genetic/environmental interactions, lead to an autism diagnosis. That is followed by a “transactional” stage, where a child’s experience with the world molds their later behavior and functioning. This view does not contradict the idea of profound autism but hypothesizes the process by which autistic individuals develop symptoms and features. It also explains the vast heterogeneity across the spectrum, why interventions and supports that are “one size fits all” are not useful and why autism is a distinct category rather than a continuation of typical variation.3
Who has autism?
This title was “borrowed” from a 2014 Day of Learning presentation given by Cathy Rice, PhD from the Centers for Disease Control. Even 10 years later, it’s a relevant question.

New prevalence numbers for autism were released by the CDC ADDM Network, which has been tracking autism prevalence for the past 20-plus years. This year the CDC reported that the number of 8-year-old children who were diagnosed with autism increased to one in 36, with variability across gender and geographic location.4 Similar data obtained from 4-year-olds show that early identification of ASD is improving, and while racial and ethnic differences still exist in the cohort of 4-year-old children,5 that disparity is reduced by 8 years of age. But while racial disparities are improving, there are still features that differentiate those from different racial backgrounds. For example, intellectual disability is still diagnosed more often in Black children. Girls are also still significantly less likely to be diagnosed than boys at 4 or 8 years of age – a difference that has been consistently demonstrated and warrants further research.
This year for the first time, the CDC also measured the prevalence of profound autism using the definition published in the Lancet in 2021 (IQ<50, minimally or nonverbal and requiring round-the-clock supports). The CDC reported that 27.8% of those with ASD meet this definition of profound autism.6 Members of this population are rarely included in research studies. In response, ASF has launched a profound autism pilot grant program specifically devoted to studying this underrepresented and understudied population.
The question of “who has autism” is muddled by increased use of social media platforms that circulate inaccurate and misleading information about autism and its features. One study showed that most of the information about autism on TikTok was inaccurate, but that these representations were viewed just as often as were accurate presentations.7 This might explain misunderstandings about autism, what causes it and what can be done to help those with a diagnosis. We will never be able to stop the spread of misinformation on the internet, but families can educate themselves about the science of autism so they know what is true and what is false.
Rage against the machine
This year saw an explosion of new technology and artificial intelligence-assisted devices to help diagnose autism.

These tools, aka “AI” technologies are being developed to better automate an autism diagnosis in the hopes of decreasing the time families spend on waiting for an evaluation.8 Many of these new AI-based projects have been reviewed elsewhere or described in the ASF Weekly Science podcast.9
Two such AI tools designed to improve diagnosis use measures of social attention and visual engagement, measuring how much time someone spends looking at social vs. nonsocial scenes. Researchers and clinicians have observed for years that children with autism tend to look less at faces and more at objects in a visual scene, and this knowledge has now been turned into screening and diagnostic tools. Duke University piloted a system called SenseToKnow, and it has shown promising results in pediatric settings.10Another technology, called EarliPoint, moved beyond the pilot stage and received FDA approval after validation for use in identifying children with autism as early as 16 months of age. Both are noninvasive and require a child to spend only a few minutes looking at different stimuli.11,12 They identify children with autism nearly as well as standard assessments, which can take hours to administer and can be costly.
Telehealth technologies like As You Are and CanvasDx (CanvasDx was FDA approved last year) are taking advantage of technological advancements to use clinician-coded measures of autism behaviors (like social communication abilities, social interaction, restrictive and repetitive behaviors, and caregiver reports of autistic behavior and impairment) to provide accessible diagnostic services. These are more like traditional assessments, but can be done in more naturalistic settings like the home over the internet. They use mathematical calculations to determine probability of a diagnosis from shorter observations.
All of these “machine learning” approaches build on existing science around what is known about the early signs of autism, which behavioral features are most salient and what can be done to minimize burden to the family and eliminate diagnostic bias, as well as provide quality assessment services in countries that have few trained experts. The goal is to reduce the unacceptable waitlists to receive an evaluation. Other machine learning approaches are being studied to identify the best method of intervention,13 sleep14 and probability of a genetic diagnosis.15 Based on the number of published research protocols, there are at least four AI-based research studies in progress to improve understanding and prediction of severe and dangerous behaviors.
These tools were built off the arsenal of scientific discovery in autism that has been established over the past decades. Without many years of eye tracking, brain tissue analysis, genetic discovery and facial expression analysis, developing tools like these would have been impossible.
Where are our girls?
With females four times less likely to receive a diagnosis for various reasons16, there is a lack of knowledge of the needs of females with autism, also called autistic females at birth.

In 2023, the Lancet published a list of suggestions for supporting females on the spectrum, ranging from recognizing “female driven” stereotyped behavior to improving employment opportunities that are traditionally focused on male interests. 17 While it is known that factors like camouflaging autistic features may hide a diagnosis, age tends to play a huge role in the features that are displayed by autistic females vs. autistic males, as well as in how these behaviors are interpreted by clinicians.18 While many non-biological hypotheses have been generated to explain the difference in diagnostic prevalence of females vs. males, there are also neurobiological markers which may explain why certain features of autism are different in females, including different activity in certain brain regions during adolescence.19,20 Scientists have been able to dissect different cells from male and female brains to confirm that female cells have a higher rate of mutations in ASD risk genes compared to males.21 Finally, more research this year points to different patterns of brain development, which may explain behavioral differences in autistic females vs. males at birth.19,20,22 Females and males are typically different, and gender disparities in diagnosis are not unique to autism. However, as females with autism are understudied and often overlooked, clinicians and care providers need more tools to help autistic females receive the support they need.
How does the autism brain work?
Understanding of basic neurological processes in autism leads to clues which influence therapeutics.

Everyone wants to know what the autism brain looks like and how it functions across the lifespan and across genders. By understanding basic neurological processes in the brain, scientists can identify tailored support options and even therapeutics. More studies this year have shown similarities in how areas of the autistic brain connect compared to brains of people with ADHD and bipolar depression. This is additional evidence that autism is part of the bigger spectrum of neurodevelopmental disorders that includes depression, schizophrenia and obsessive-compulsive disorder. It also indicates that, while the autism brain may have a distinct profile, it has many similarities to other conditions and disorders.23
Therapeutic clues are being identified from circuits and molecular markers in the brain. By studying brain tissue, scientists have found that genes controlling how neurons connect and communicate are dampened in autism, while those affecting inflammation are heightened. This finding has been confirmed in other body tissue24 and model systems.25 The changes suggest that initial overexcitation and overconnectivity of cells in the brain lead to inflammation and potential neurodegeneration. These changes may worsen over time, opening up areas of research not just in childhood but through adulthood.26 One of the genes of interest, GABA, turns cells off and is thought of as a “traffic light,” ensuring that not all the lights are green and that traffic flows safely. Brain tissue studies revealed that a decrease in the number of cells that contain GABA (and thus ensure that cells do not become too excited) is linked to two specific features of ASD: stereotypy and intellectual ability.27 The study of brain tissue and the ability to look directly at the brains of people with autism is only made possible by families who have learned more about the benefits of postmortem brain donation through the Autism BrainNet.
Finally, new model systems, called assembloids, are also now being used to identify and understand not just the genes associated with autism, but how they function in cells.28 By using assembloids, sometimes referred to as “brains in a dish,” hundreds of different genes can be systematically altered and the functions of the cells examined to determine what those genes do. Using these systems, researchers found that a gene associated with ASD affected the function of a part of the cell called the endoplasmic reticulum, which resulted in abnormal migration, i.e., brain cells did not go to the right places during development.29
Cha-Cha-Cha-Changes
Families and individuals experience autism across the lifespan, so why should it just be studied at one time point?



Longitudinal studies that track individuals over time are rare but critical in developing our understanding of autism trajectories. Studies this year point to at least three groups of trajectories: kids who start off with fewer signs and continue to have relatively fewer support needs; kids who are more impaired and stay relatively stable; and kids whose symptoms worsen over time. Features at 6 months were able to predict outcome at 5-6 years with some (but not absolute) accuracy.30 While some studies point to three or four trajectory “groups,” others show at least six.31
Longitudinal studies also indicated that people with autism show higher levels of mental health problems across childhood, predicted by IQ and gender, 32 with girls showing higher levels of depression and anxiety. Higher IQ at age 2 predicted lower levels of attention problems.32 From midlife through early old age, physical health worsened (as expected) but autism symptoms improved, with differences seen between those with and without intellectual disability.33 Other “turning points” or critical windows for change seem to happen in childhood and early adolescence,34 reinforcing that change over time is not linear; there are many peaks and valleys along the way, influenced again by intellectual ability. Females seem to be disproportionately affected by medical and psychiatric issues when transitioning from adolescence into adulthood.35 Future studies should integrate core autism features as well as comorbid conditions36 to better understand the abilities, disabilities and trajectories of those with ASD.
Genetics is not always deterministic
New understanding of the types of gene changes, where they occur in the body and how they influence development can influence the type of treatment a person receives.
There has been great debate over the utility of genetic research in ASD, including the roles of different types of gene mutations in functioning and outcome. Even among rare genetic syndromes associated with autism, there is great heterogeneity in features both within a diagnosis,37,38 and across disorders.39 So how deterministic are single gene mutations associated with autism? Recently, Rolland and colleagues demonstrated that some individuals who had rare genetic mutations did not qualify for a diagnosis,40 sparking debate about resilience to genetic mutations. Is it social environment, epigenetic factors, gender or other genes in the background that are conferring protection?
Recent evidence suggests that assortative mating may explain rare genetic variants in children with rare genetic forms of autism.41 The phenotypic similarities of the biological parents have only before been hypothesized for common variation, but this new work suggests a role for rare variation as well. In addition, those with rare genetic mutations and ASD are also more likely to have common variants, each with their own distinct influence on brain development and autism behaviors. This is consistent with a liability threshold model, where family members who may share genetic risk factors do not have a diagnosis, but where the rare genetic and common genetic variants add to a broad continuum of traits. In some of these cases, it can lead to a diagnosis.42 More genetic research will help identify how rare and common gene variants interact, how they lead to an autism diagnosis, what neural mechanisms are involved and whether and how the genes lead to other outcomes besides autism.
Sometimes it’s not all about the autism
Comorbidites of autism often are life-threatening and impair quality of life. There are some solutions.

Comorbid medical and psychiatric issues are a prescient issue in the autism community because individuals with ASD experience a higher burden of these disorders and conditions.43 There is an enduring recognition that comorbidities of autism can be severe, impair daily life and, in the case of catatonia and seizures, even be fatal. Some features associated with these comorbidities can be explained by etiological factors like family history and preterm birth.43 Many genes that are related to autism have multiple functions all over the body, including the GI system, leading to GI distress and dysfunction with an autism diagnosis.44 Identification of these genes has led to potential therapies.45-47 Most comorbidities are present across the lifespan, while some improve over time.48
The unspoken heroes of autism families: siblings!
Siblings have a unique relationship that lasts an entire life. Who knows you better than your sister or brother?

Research shows that nonautistic siblings of people with autism are at higher risk for psychiatric and neurodevelopmental disorders compared to the general population. A thorough review of the literature published this year indicates a decreased quality of life and sense of well-being in non-autistic siblings. This varied by age, sibling relationship and other factors, but was found to be consistent across studies.49 There is not yet enough research to draw any conclusion about psychopathology in siblings, although depression was detected in some small studies.50 A new intervention incorporating cognitive behavioral therapy, positive psychology and stress management showed promise in increasing resilience to adverse events as well as promoting improved coping around stressful events in siblings.51
The presence of a typically developing sibling has a beneficial effect on a multitude of outcomes for their autistic brother or sister, although differences in ages, sibling relationships and presence of challenging behaviors influenced the strength of the findings.52 The IQ of the autistic sibling and the presence of externalizing behaviors significantly influenced conflict between siblings. Instances where the proband had an intellectual disability showed the lowest level of conflict, while instances where the proband was prone to aggression resulted in the highest levels of conflict.53
Final thoughts
After a diagnosis, the first task is to find appropriate services, supports and interventions. But what works in one person does not necessarily work in another.
The goal of autism research is to help individuals with autism and their families lead their best lives. The rise of personalized medicine will improve opportunities to deliver the right treatments to the right person at the right time. Gene-first studies are identifying treatments that are targeted at underlying biological causes in individuals with monogenic forms of autism. Longitudinal studies can identify critical windows for intervention. AI-driven technologies are being developed to improve diagnosis and mitigate symptoms.12 Comprehensive meta-analyses continue to reinforce the benefits of behavioral and developmental interventions for core features of autism. 54 And studies continue to confirm that “earlier is better” when it comes to intervention. For example, a new randomized clinical trial confirmed that 18-to-24-month-old children who received more intensive, individualized forms of intervention showed greater gains in language, social communication and self-help than those who received intervention later.55 Another study showed that those with better baseline adaptive skills had such strong responses to early intervention that their diagnosis was no longer evident by school age, suggesting this may influence response to intervention.56 Research this year also demonstrated that meaningful gains in language can be made in autistic children who are non-speaking through behavioral and naturalistic developmental interventions,57 as well as confirming that early intervention saves costs in the long term.58,59 No single intervention is going to support everyone across the autism spectrum equally, but it is clear that access to early, evidence-based intervention to help children live their best lives possible is imperative for child health and development.
References
1. Green J. Debate: Neurodiversity, autism and healthcare. Child Adolesc Ment Health 2023; 28(3): 438-42.
2. Whitehouse A. Commentary: A spectrum for all? A response to Green et al. (2023), neurodiversity, autism and health care. Child Adolesc Ment Health 2023; 28(3): 443-5.
3. Frazier TW, Chetcuti L, Al-Shaban FA, et al. Categorical versus dimensional structure of autism spectrum disorder: A multi-method investigation. JCPP Adv 2023; 3(2): e12142.
4. Maenner MJ, Warren Z, Williams AR, et al. Prevalence and Characteristics of Autism Spectrum Disorder Among Children Aged 8 Years – Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill Summ 2023; 72(2): 1-14.
5. Shaw KA, Bilder DA, McArthur D, et al. Early Identification of Autism Spectrum Disorder Among Children Aged 4 Years – Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2020. MMWR Surveill Summ 2023; 72(1): 1-15.
6. Hughes MM, Shaw KA, DiRienzo M, et al. The Prevalence and Characteristics of Children With Profound Autism, 15 Sites, United States, 2000-2016. Public Health Rep 2023; 138(6): 971-80.
7. Aragon-Guevara D, Castle G, Sheridan E, Vivanti G. The Reach and Accuracy of Information on Autism on TikTok. J Autism Dev Disord 2023.
8. Cognoa I. The State of Pediatric Autism Diagnosis in the U.S.:
Gridlocks, Inequities and Missed Opportunities Persist. https://cognoa.com/waitlist-crisis-report/, 2023.
9. Aylward BS, Abbas H, Taraman S, et al. An Introduction to Artificial Intelligence in Developmental and Behavioral Pediatrics. J Dev Behav Pediatr 2023; 44(2): e126-e34.
10. Perochon S, Di Martino JM, Carpenter KLH, et al. Early detection of autism using digital behavioral phenotyping. Nat Med 2023; 29(10): 2489-97.
11. Jones W, Klaiman C, Richardson S, et al. Eye-Tracking-Based Measurement of Social Visual Engagement Compared With Expert Clinical Diagnosis of Autism. JAMA 2023; 330(9): 854-65.
12. Jones W, Klaiman C, Richardson S, et al. Development and Replication of Objective Measurements of Social Visual Engagement to Aid in Early Diagnosis and Assessment of Autism. JAMA Netw Open 2023; 6(9): e2330145.
13. Twala B, Molloy E. On effectively predicting autism spectrum disorder therapy using an ensemble of classifiers. Sci Rep 2023; 13(1): 19957.
14. Eylon G, Tikotzky L, Dinstein I. Performance evaluation of Fitbit Charge 3 and actigraphy vs. polysomnography: Sensitivity, specificity, and reliability across participants and nights. Sleep Health 2023; 9(4): 407-16.
15. Donnelly N, Cunningham A, Salas SM, et al. Identifying the neurodevelopmental and psychiatric signatures of genomic disorders associated with intellectual disability: a machine learning approach. Mol Autism 2023; 14(1): 19.
16. McFayden TC, Putnam O, Grzadzinski R, Harrop C. Sex Differences in the Developmental Trajectories of Autism Spectrum Disorder. Curr Dev Disord Rep 2023; 10(1): 80-91.
17. Lai MC, Amestoy A, Bishop S, et al. Improving autism identification and support for individuals assigned female at birth: clinical suggestions and research priorities. Lancet Child Adolesc Health 2023; 7(12): 897-908.
18. Tsirgiotis JM, Young RL, Weber N. A comparison of the presentations of males and females with autism spectrum disorder and those narrowly below the diagnostic threshold. Autism 2023: 13623613231190682.
19. Neuhaus E, Santhosh M, Kresse A, et al. Frontal EEG alpha asymmetry in youth with autism: Sex differences and social-emotional correlates. Autism Res 2023.
20. St John T, Estes AM, Hazlett HC, et al. Association of Sex With Neurobehavioral Markers of Executive Function in 2-Year-Olds at High and Low Likelihood of Autism. JAMA Netw Open 2023; 6(5): e2311543.
21. Velmeshev D, Perez Y, Yan Z, et al. Single-cell analysis of prenatal and postnatal human cortical development. Science 2023; 382(6667): eadf0834.
22. Gros G, Miranda Marcos R, Latrille A, et al. Whole-brain gray matter maturation trajectories associated with autistic traits from adolescence to early adulthood. Brain Struct Funct 2023.
23. Segal A, Parkes L, Aquino K, et al. Regional, circuit and network heterogeneity of brain abnormalities in psychiatric disorders. Nat Neurosci 2023; 26(9): 1613-29.
24. Dumitriu D, Baldwin E, Coenen RJJ, et al. Deciduous tooth biomarkers reveal atypical fetal inflammatory regulation in autism spectrum disorder. iScience 2023; 26(3): 106247.
25. Weinschutz Mendes H, Neelakantan U, Liu Y, et al. High-throughput functional analysis of autism genes in zebrafish identifies convergence in dopaminergic and neuroimmune pathways. Cell Rep 2023; 42(3): 112243.
26. Zhang P, Omanska A, Ander BP, Gandal MJ, Stamova B, Schumann CM. Neuron-specific transcriptomic signatures indicate neuroinflammation and altered neuronal activity in ASD temporal cortex. Proc Natl Acad Sci U S A 2023; 120(10): e2206758120.
27. Dufour BD, McBride E, Bartley T, Juarez P, Martinez-Cerdeno V. Distinct patterns of GABAergic interneuron pathology in autism are associated with intellectual impairment and stereotypic behaviors. Autism 2023; 27(6): 1730-45.
28. Wang L, Owusu-Hammond C, Sievert D, Gleeson JG. Stem Cell-Based Organoid Models of Neurodevelopmental Disorders. Biol Psychiatry 2023; 93(7): 622-31.
29. Meng X, Yao D, Imaizumi K, et al. Assembloid CRISPR screens reveal impact of disease genes in human neurodevelopment. Nature 2023; 622(7982): 359-66.
30. Franchini M, Smith IM, Sacrey L, et al. Continuity of trajectories of autism symptom severity from infancy to childhood. J Child Psychol Psychiatry 2023; 64(6): 895-906.
31. Fountain C, Winter AS, Cheslack-Postava K, Bearman PS. Developmental Trajectories of Autism. Pediatrics 2023; 152(3).
32. Wright N, Courchesne V, Pickles A, et al. A longitudinal comparison of emotional, behavioral and attention problems in autistic and typically developing children. Psychol Med 2023: 1-13.
33. Hong J, DaWalt LS, Taylor JL, Haider A, Mailick M. Autism through midlife: trajectories of symptoms, behavioral functioning, and health. J Neurodev Disord 2023; 15(1): 36.
34. Chen Y-J, Duku E, Szatmari P, et al. Trajectories of adaptive functioning from early childhood to adolescence in autism: Identifying turning points and key correlates of chronogeneity. JCPP Advances 2023; n/a(n/a): e12212.
35. Malow BA, Qian Y, Ames JL, Alexeeff S, Croen LA. Health conditions in autism: Defining the trajectory from adolescence to early adulthood. Autism Res 2023; 16(7): 1437-49.
36. Waizbard-Bartov E, Fein D, Lord C, Amaral DG. Autism severity and its relationship to disability. Autism Res 2023; 16(4): 685-96.
37. Bozhilova N, Welham A, Adams D, et al. Profiles of autism characteristics in thirteen genetic syndromes: a machine learning approach. Mol Autism 2023; 14(1): 3.
38. Jenner L, Richards C, Howard R, Moss J. Heterogeneity of Autism Characteristics in Genetic Syndromes: Key Considerations for Assessment and Support. Curr Dev Disord Rep 2023; 10(2): 132-46.
39. Peall KJ, Owen MJ, Hall J. Rare genetic brain disorders with overlapping neurological and psychiatric phenotypes. Nature Reviews Neurology 2023.
40. Rolland T, Cliquet F, Anney RJL, et al. Phenotypic effects of genetic variants associated with autism. Nat Med 2023; 29(7): 1671-80.
41. Smolen C, Jensen M, Dyer L, et al. Assortative mating and parental genetic relatedness contribute to the pathogenicity of variably expressive variants. The American Journal of Human Genetics 2023; 110(12): 2015-28.
42. Cirnigliaro M, Chang TS, Arteaga SA, et al. The contributions of rare inherited and polygenic risk to ASD in multiplex families. Proc Natl Acad Sci U S A 2023; 120(31): e2215632120.
43. Khachadourian V, Mahjani B, Sandin S, et al. Comorbidities in autism spectrum disorder and their etiologies. Transl Psychiatry 2023; 13(1): 71.
44. Davidson EA, Holingue C, Jimenez-Gomez A, Dallman JE, Moshiree B. Gastrointestinal Dysfunction in Genetically Defined Neurodevelopmental Disorders. Semin Neurol 2023; 43(4): 645-60.
45. Kim HK, Goncalves VF, Husain MI, et al. Cross-disorder GWAS meta-analysis of endocannabinoid DNA variations in major depressive disorder, bipolar disorder, attention deficit hyperactivity disorder, autism spectrum disorder, and schizophrenia. Psychiatry Res 2023; 330: 115563.
46. Lai MC. Mental health challenges faced by autistic people. Nat Hum Behav 2023; 7(10): 1620-37.
47. Curatolo P, Scheper M, Emberti Gialloreti L, Specchio N, Aronica E. Is tuberous sclerosis complex-associated autism a preventable and treatable disorder? World J Pediatr 2023.
48. Micai M, Fatta LM, Gila L, et al. Prevalence of co-occurring conditions in children and adults with autism spectrum disorder: A systematic review and meta-analysis. Neurosci Biobehav Rev 2023; 155: 105436.
49. Quatrosi G, Genovese D, Amodio E, Tripi G. The Quality of Life among Siblings of Autistic Individuals: A Scoping Review. J Clin Med 2023; 12(3).
50. Bispo-Torres AC, Lucena R, Tavares-Rodrigues IC, et al. Psychopathological symptoms in parents and siblings of people on the autism spectrum: A systematic review and meta-analysis. Psychiatry Res2023; 323: 115145.
51. Kuhlthau KA, Traeger L, Luberto CM, et al. Resiliency Intervention for Siblings of Children With Autism Spectrum Disorder: A Randomized Pilot Trial. Acad Pediatr 2023; 23(6): 1187-95.
52. Cuskelly M, Gilmore L, Rayner C, Girkin F, Mulvihill A, Slaughter V. The impacts of typically developing siblings on the developmental outcomes of children with disability: A scoping review. Res Dev Disabil 2023; 140: 104574.
53. Rixon L, Hastings RP, Kovshoff H, Bailey T. Short Report: Outcomes for siblings associated with sub-groups of autistic children with intellectual disability identified by latent profile analysis. Res Dev Disabil2022; 130: 104337.
54. Sandbank M, Bottema-Beutel K, Crowley LaPoint S, et al. Autism intervention meta-analysis of early childhood studies (Project AIM): updated systematic review and secondary analysis. BMJ 2023; 383: e076733.
55. Guthrie W, Wetherby AM, Woods J, et al. The earlier the better: An RCT of treatment timing effects for toddlers on the autism spectrum. Autism 2023; 27(8): 13623613231159153.
56. Harstad E, Hanson E, Brewster SJ, et al. Persistence of Autism Spectrum Disorder From Early Childhood Through School Age. JAMA Pediatr 2023; 177(11): 1197-205.
57. Kasari C, Shire S, Shih W, Landa R, Levato L, Smith T. Spoken language outcomes in limited language preschoolers with autism and global developmental delay: RCT of early intervention approaches. Autism Res 2023; 16(6): 1236-46.
58. Tinelli M, Roddy A, Knapp M, et al. Economic analysis of early intervention for autistic children: findings from four case studies in England, Ireland, Italy, and Spain. Eur Psychiatry 2023; 66(1): e76.
59. Segal L, Green J, Twizeyemariya A, et al. Estimated Therapy Costs and Downstream Cost Consequences of iBASIS-Video Interaction to Promote Positive Parenting Intervention vs Usual Care Among Children Displaying Early Behavioral Signs of Autism in Australia. JAMA Netw Open 2023; 6(4): e235847.
The post 2023 Year End Review appeared first on Autism Science Foundation.
]]>The post Autism Updates: ASF Goes to INSAR appeared first on Autism Science Foundation.
]]>Read about what ASF presented and discovered for autism updates at INSAR 2023.
Autism Does Not Mean The Same Thing to Every Person
This was a theme throughout INSAR. For some, autism is an identity, others a condition, and others a disorder. Everyone with autism is different, genetically, sociologically, demographically. What is true of one person, even one autistic person, is not true of another. Everyone’s voice matters, but we should be doing better about including autistic voices in scientific research.
Some voices from the autism community have expressed concerns about the scientific study of autism. One roundtable discussion group of neurodiverse autistic people centered around the feeling that genetic studies will be used to eliminate people with autism. These feelings linger despite genetic studies having the clearly defined purpose of helping with autism diagnosis and treatment.
There is so much to learn from the study of genetics. Like the community itself, autism is diverse and complex. Genetic studies have helped doctors be able to recommend treatments for families with autism. The largest genetic study to date found insights into some of the biological complexities involved in autism, which can help develop new approaches to diagnosis and treatment.
As ASF Chief Science Officer Dr. Alycia Halladay said on her ASF podcast, we need to work together as a community with interactions that are not solely based in fear and anger. We can be careful about how genetics is used. But we don’t need to stop science from being done. There are a multitude of conditions that are autism. There are so many discoveries to be made that will help with diagnosis and treatment. More studies are required, not less.

Improve Accessibility for Participants in Autism Research
Minimally verbal or intellectually disabled people are being excluded from autism research. One of the INSAR panels seeking to address this problem was “Breaking Down Barriers to Research for Children with Genetic Disorders, Intellectual Disability or Communication Challenges.“
The goal of the panel was to share methods of helping participants to ensure families can complete the assessments to help their children. The chair, Dr. Carol Wilkinson, used a Swiss cheese analogy to show that participation can’t always follow a straight line. For example, some families get started but circumstances prevent them from finishing. Or they can’t get to the clinic to start. Or they complete biological assessments but then they can’t collect IQ.
So what are the recommendations to help improve research participation from families?
- Be flexible. Tests and protocols likely will need to be adapted for each participant. Many children on the spectrum have sensory issues. Some have challenging behaviors. Others may not understand what they have to do to participate. Researchers should look at the the difference in developmental level ratio against chronological age. In other words, don’t assume what participants can or can’t do based on their age. Skip unnecessary and non-applicable material as needed.
- Meet them where they are at. Collect data in the most naturalistic setting where the participant feels most comfortable. Even if it means leaving your own comfortable place. Dr. Caitlin Hudac from the University of South Carolina shared how she spent 92 days on the road, collecting data from families within their own surroundings.
- Use a personalized approach. Get as much information about your participant(s) ahead of time as possible. Examples: what’s their favorite toy, game, snack, etc.? Getting to know the people involved in research helps the family participate and improves the quality of data.
- Plan for remote. It’s not always feasible to meet in-person with families during research. But that’s not the only way to do assessments or collect data. Dr. Karen Chenausky completed many parts of her research over Zoom, including language assessments, parent-child interactions, and motor tasks. She was able to protect the participants’ privacy and use a free downloadable app that parents used to measure language.

Autism and Aging
Internationally renowned expert Patricia Howlin from Kings College London talked about adulthood with autism. She has studied autism from birth to adulthood, seen autistic kids grow to be adults, and has used a unique longitudinal study design to address what happens to autistic kids as they get old. Unfortunately, there have not been enough studies in this area, the existing studies are designed differently, and not enough people are in each study.
We still need multiple approaches, but people with autism as they age don’t have nearly the amount of information about them as those young children. That should change. The dynamics and needs of people aging with autism are as a varied as the spectrum itself. Additional studies should be expanded with the goal of determining precise and diverse definitions of quality of life for autistic people as they age.

Autism in Females
Girls are an underserved community in autism that needs more support and better diagnostics. This was made clear through multiple presentations at INSAR. To partially illustrate, this video from SPARK for Autism shows some of the needs specific to women and girls.
Studies show higher burdens of rare genes and common genes associated with ASD and non-diagnosed family members like mothers or sisters. This includes a South Korea study which replicated other studies with samples from around the world. There are many other traits unique to females with autism that warrant further study, including what’s called the “female protective effect” which theorizes that women and girls are better able to tolerate known genetic mutations. Multiple studies support this theory. It may help explain autism being less common among females.
Research has also shown that females with ASD are diagnosed later and tend to have a lower IQ. But a couple of subsets of children deserve a closer look to investigate what may be part of the female protective effect. These children either showed autistic traits early in life but then didn’t meet autism criteria later, or they met autism criteria in toddlerhood but didn’t meet the diagnostic threshold at a later school age. Girls in the first group (early life autistic traits that didn’t increase with age) showed poorer language ability. Girls from the second subset (those who missed the early diagnosis) had better language and less pronounced autism features but more problems in peer relationships. Are either of these related to the female protective effect? We don’t have a clear answer yet.
Utilize The Autistic Community to Broaden Perspective
How do we work with the autistic community to conduct science that is important but at the same time communicate the importance of that science? One of INSAR’s advocate panels talked about what should be done and not done using Spectrum10k as an example. After backlash and concerns about the study design and principal investigators, the study’s leaders went back to the drawing board. They created and documented a new process. It was made for those that believe in genetics research as well as those in the community that are fearful of it.
While there is no exact recipe, including the autism community in research should be mandatory and based on the following principles:
- Trustworthiness; everything should be transparent, clear, and balanced.
- Autistic-led and based on community priorities, not just the loudest voices on social media.
- Include a diversity of voices and use engagement methods that are accessible. Provide physical and language accommodations as needed, such as type to text.
- Ensure that something gets done, not just talked about, as part of the consultation.
Autism science and research should benefit the larger community. Including a broad range of perspectives is critical.
INSAR Videos On-Demand
Videos from INSAR plenary sessions will be available starting June 5. This on-demand content will only be available until July 10 but is free to INSAR members. INSAR Membership reopens June 1.
The post Autism Updates: ASF Goes to INSAR appeared first on Autism Science Foundation.
]]>The post Autism Updates: Profound Autism Child Mind Institute, INSAR 2023, DOL Highlights Video appeared first on Autism Science Foundation.
]]>
Profound Autism Article from the Child Mind Institute
“The term profound is not meant to demean anyone or label anyone as a lost cause,” said ASF President Alison Singer. “Just the opposite. The whole idea of creating this nomenclature is so we are better prepared to meet the needs of this population and not pretend they don’t exist.”
“The main purpose was to call attention to the fact that these kids and adults exist, and that they do need different services,” said Catherine Lord, PhD, the co-chair of the Lancet Commission.
Read the full story: https://childmind.org/article/what-is-profound-autism/
INSAR 2023
ASF President Alison Singer and Chief Science Officer Dr. Alycia Halladay are in Stockholm this week as attendees and presenters for the 2023 International Society of Autism Research annual meeting. More highlights to come soon!



2023 Day of Learning Highlights
Watch a video recap of the 2023 Day of Learning. You can watch all the presentations from every speaker on our YouTube channel.
The post Autism Updates: Profound Autism Child Mind Institute, INSAR 2023, DOL Highlights Video appeared first on Autism Science Foundation.
]]>